Johnson and johnson vaccine trial information
Home » » Johnson and johnson vaccine trial informationYour Johnson and johnson vaccine trial images are ready in this website. Johnson and johnson vaccine trial are a topic that is being searched for and liked by netizens today. You can Download the Johnson and johnson vaccine trial files here. Find and Download all free images.
If you’re looking for johnson and johnson vaccine trial images information related to the johnson and johnson vaccine trial interest, you have visit the ideal site. Our website frequently gives you suggestions for refferencing the maximum quality video and picture content, please kindly hunt and find more enlightening video articles and graphics that match your interests.
Johnson And Johnson Vaccine Trial. Janssen All of the COVID-19 vaccines come with the possibility of side effects. It took AstraZeneca researchers 1 month to get vaccine trial data to FDA. Johnson Johnson and AstraZeneca will resume their coronavirus vaccine trials in the US after pauses to investigate unexplained illnesses in trial participants. New Brunswick NJ April 2 2021 Johnson Johnson the Company has begun vaccinating adolescent participants in the ongoing Phase 2a clinical trial for its COVID-19 vaccine candidate developed by the Janssen Pharmaceutical Companies of Johnson Johnson.
 Massachusetts Explores The Advantages And Potential Challenges Of The Johnson Johnson Vaccine Commonhealth From wbur.org
Massachusetts Explores The Advantages And Potential Challenges Of The Johnson Johnson Vaccine Commonhealth From wbur.org
Johnson Johnson Covid-19 vaccine is 66 effective in global trial but 85 effective against severe disease company says. People had the most protection 2 weeks after getting vaccinated. Johnson Johnson NYSEJNJ announced that its single-shot Covid-19 vaccine was 66 effective in preventing moderate and severe cases of Covid-19 in its global phase 3 trials. The FDA granted the JJ COVID-19 vaccine emergency use. The COVID-19 vaccine from Johnson Johnson uses existing technology that involves a virus called adenovirus a common cause of respiratory infections. Johnson Johnsons Phase III double-dose Covid-19 vaccine prompted a range of expert analyses as to how the prime-boost regimen would be rolled out considering the single-dose approach is already available in the US.
The COVID-19 vaccine from Johnson Johnson uses existing technology that involves a virus called adenovirus a common cause of respiratory infections.
The JJJanssen COVID-19 Vaccine was 663 effective in clinical trials efficacy at preventing laboratory-confirmed COVID-19 infection in people who received the vaccine and had no evidence of being previously infected. The agency reviewed clinical trial data from over 40000 participants as well as manufacturing information that Johnson and Johnson provided. Early Johnson Johnson COVID-19 vaccine trial data shows antibody response after 8 days. Johnson Johnson and the FDA also declined to answer whether this was the first pause for its Covid-19 vaccine trial. Our knowledge and experience successes and set-backs alike inform everything were doing today to help eradicate COVID-19. Johnson Johnson Statement on COVID-19 Vaccine.
 Source: cnbc.com
Source: cnbc.com
Johnson Johnson and the FDA also declined to answer whether this was the first pause for its Covid-19 vaccine trial. Johnson Johnson Statement on COVID-19 Vaccine. For over a century Johnson Johnson has actively engaged in fighting global outbreaks from TB to HIV and Ebola. Johnson Johnson expands Covid-19 vaccine trial to adolescents 05 Apr 2021 Last Updated April 6th 2021 1005 Johnson Johnson JJ has expanded Phase IIa clinical trial of Covid-19 vaccine candidate to include adolescent subjects aged 12 to 17 years. The JJJanssen COVID-19 Vaccine was 663 effective in clinical trials efficacy at preventing laboratory-confirmed COVID-19 infection in people who received the vaccine and had no evidence of being previously infected.
 Source: jnj.com
Source: jnj.com
Johnson Johnsons Phase III double-dose Covid-19 vaccine prompted a range of expert analyses as to how the prime-boost regimen would be rolled out considering the single-dose approach is already available in the US. In the JJ vaccine clinical trial 2650 trial participants were tested using a COVID-19 antigen test 71 days after vaccination. A doctor administers AstraZenecas coronavirus vaccine as part of a clinical trial in São Paulo Brazil on July 24 2020. The JJJanssen COVID-19 Vaccine was 663 effective in clinical trials efficacy at preventing laboratory-confirmed COVID-19 infection in people who received the vaccine and had no evidence of being previously infected. For over a century Johnson Johnson has actively engaged in fighting global outbreaks from TB to HIV and Ebola.
 Source: npr.org
Source: npr.org
The COVID-19 vaccine from Johnson Johnson uses existing technology that involves a virus called adenovirus a common cause of respiratory infections. Johnson Johnson Statement on COVID-19 Vaccine. That found that the AstraZeneca vaccine which uses similar technology as Johnson Johnson has a 92 effectiveness against hospitalization after two. JJs single-dose approach would remain authorised even if the two-dose approach is authorised. Johnson Johnson expands Covid-19 vaccine trial to adolescents 05 Apr 2021 Last Updated April 6th 2021 1005 Johnson Johnson JJ has expanded Phase IIa clinical trial of Covid-19 vaccine candidate to include adolescent subjects aged 12 to 17 years.
 Source: dw.com
Source: dw.com
Fauci cited a study from the UK. People had the most protection 2 weeks after getting vaccinated. Johnson Johnson NYSEJNJ announced that its single-shot Covid-19 vaccine was 66 effective in preventing moderate and severe cases of Covid-19 in its global phase 3 trials. A clinical trial volunteer participates in Johnson Johnsons study to test a coronavirus vaccine. Johnson Johnson expands Covid-19 vaccine trial to adolescents 05 Apr 2021 Last Updated April 6th 2021 1005 Johnson Johnson JJ has expanded Phase IIa clinical trial of Covid-19 vaccine candidate to include adolescent subjects aged 12 to 17 years.
 Source: nbcnews.com
Source: nbcnews.com
A clinical trial volunteer participates in Johnson Johnsons study to test a coronavirus vaccine. Johnson Johnson Covid-19 vaccine is 66 effective in global trial but 85 effective against severe disease company says. In the JJ vaccine clinical trial 2650 trial participants were tested using a COVID-19 antigen test 71 days after vaccination. Johnson Johnson expands Covid-19 vaccine trial to adolescents 05 Apr 2021 Last Updated April 6th 2021 1005 Johnson Johnson JJ has expanded Phase IIa clinical trial of Covid-19 vaccine candidate to include adolescent subjects aged 12 to 17 years. Johnson Johnson NYSEJNJ announced that its single-shot Covid-19 vaccine was 66 effective in preventing moderate and severe cases of Covid-19 in its global phase 3 trials.
 Source: nbcnews.com
Source: nbcnews.com
JNJ is trailing the leaders in the coronavirus vaccine race but the healthcare giant is approaching the finish line thanks to cutting the size of its phase 3 clinical. New Brunswick NJ April 2 2021 Johnson Johnson the Company has begun vaccinating adolescent participants in the ongoing Phase 2a clinical trial for its COVID-19 vaccine candidate developed by the Janssen Pharmaceutical Companies of Johnson Johnson. Johnson Johnson and AstraZeneca will resume their coronavirus vaccine trials in the US after pauses to investigate unexplained illnesses in trial participants. A doctor administers AstraZenecas coronavirus vaccine as part of a clinical trial in São Paulo Brazil on July 24 2020. In the JJ vaccine clinical trial 2650 trial participants were tested using a COVID-19 antigen test 71 days after vaccination.
 Source: mcgill.ca
Source: mcgill.ca
The expansion of Johnson Johnsons COVID-19 vaccine trial is Phase 2a of the companys clinical trial. Janssen Vaccines and Prevention one of the Janssen Pharmaceuticals companies acquired by Johnson Johnson was the regulatory sponsor of the trial and holder of the Investigational New Drug. JJs single-dose approach would remain authorised even if the two-dose approach is authorised. The FDA granted the JJ COVID-19 vaccine emergency use. Johnson Johnson Statement on COVID-19 Vaccine.
 Source: pharmaceutical-technology.com
Source: pharmaceutical-technology.com
Johnson Johnson NYSE. Janssen All of the COVID-19 vaccines come with the possibility of side effects. JJs single-dose approach would remain authorised even if the two-dose approach is authorised. Johnson Johnson Statement on COVID-19 Vaccine. Our knowledge and experience successes and set-backs alike inform everything were doing today to help eradicate COVID-19.
 Source: gavi.org
Source: gavi.org
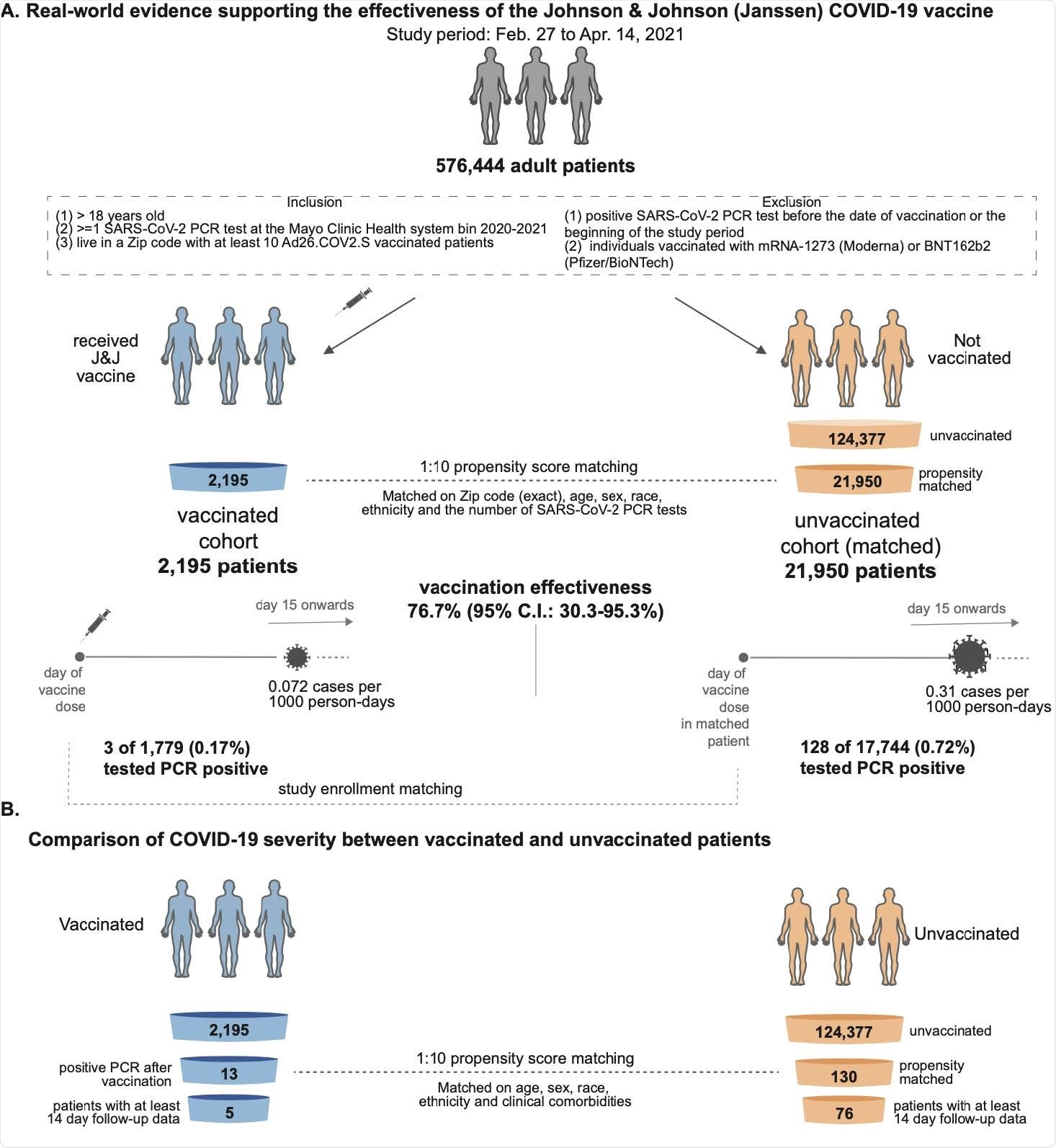
Johnson Johnson and the FDA also declined to answer whether this was the first pause for its Covid-19 vaccine trial. The DNA in the adenovirus is modified so that it produces a key part of the SARS-CoV-2 virus particle to which the body then develops an immune response. A doctor administers AstraZenecas coronavirus vaccine as part of a clinical trial in São Paulo Brazil on July 24 2020. The agency reviewed clinical trial data from over 40000 participants as well as manufacturing information that Johnson and Johnson provided. Johnson Johnson Statement on COVID-19 Vaccine.
 Source: news-medical.net
Source: news-medical.net
Johnson Johnson and the FDA also declined to answer whether this was the first pause for its Covid-19 vaccine trial. New Brunswick NJ April 2 2021 Johnson Johnson the Company has begun vaccinating adolescent participants in the ongoing Phase 2a clinical trial for its COVID-19 vaccine candidate developed by the Janssen Pharmaceutical Companies of Johnson Johnson. Johnson Johnson NYSEJNJ announced that its single-shot Covid-19 vaccine was 66 effective in preventing moderate and severe cases of Covid-19 in its global phase 3 trials. Janssen Vaccines and Prevention one of the Janssen Pharmaceuticals companies acquired by Johnson Johnson was the regulatory sponsor of the trial and holder of the Investigational New Drug. Johnson Johnson expands Covid-19 vaccine trial to adolescents 05 Apr 2021 Last Updated April 6th 2021 1005 Johnson Johnson JJ has expanded Phase IIa clinical trial of Covid-19 vaccine candidate to include adolescent subjects aged 12 to 17 years.
 Source: jnj.com
Source: jnj.com
People had the most protection 2 weeks after getting vaccinated. The FDA granted the JJ COVID-19 vaccine emergency use. Janssen Vaccines and Prevention one of the Janssen Pharmaceuticals companies acquired by Johnson Johnson was the regulatory sponsor of the trial and holder of the Investigational New Drug. Our knowledge and experience successes and set-backs alike inform everything were doing today to help eradicate COVID-19. JNJ is trailing the leaders in the coronavirus vaccine race but the healthcare giant is approaching the finish line thanks to cutting the size of its phase 3 clinical.
 Source: washingtonpost.com
Source: washingtonpost.com
Janssen All of the COVID-19 vaccines come with the possibility of side effects. Johnson Johnson NYSEJNJ announced that its single-shot Covid-19 vaccine was 66 effective in preventing moderate and severe cases of Covid-19 in its global phase 3 trials. People had the most protection 2 weeks after getting vaccinated. Johnson Johnson Covid-19 vaccine is 66 effective in global trial but 85 effective against severe disease company says. A doctor administers AstraZenecas coronavirus vaccine as part of a clinical trial in São Paulo Brazil on July 24 2020.
 Source: statnews.com
Source: statnews.com
Our knowledge and experience successes and set-backs alike inform everything were doing today to help eradicate COVID-19. Early Johnson Johnson COVID-19 vaccine trial data shows antibody response after 8 days. Janssen All of the COVID-19 vaccines come with the possibility of side effects. Fauci cited a study from the UK. The agency reviewed clinical trial data from over 40000 participants as well as manufacturing information that Johnson and Johnson provided.
 Source: wbur.org
Source: wbur.org
New Brunswick NJ April 2 2021 Johnson Johnson the Company has begun vaccinating adolescent participants in the ongoing Phase 2a clinical trial for its COVID-19 vaccine candidate developed by the Janssen Pharmaceutical Companies of Johnson Johnson. Fauci cited a study from the UK. The JJJanssen COVID-19 Vaccine was 663 effective in clinical trials efficacy at preventing laboratory-confirmed COVID-19 infection in people who received the vaccine and had no evidence of being previously infected. Janssen All of the COVID-19 vaccines come with the possibility of side effects. The expansion of Johnson Johnsons COVID-19 vaccine trial is Phase 2a of the companys clinical trial.
 Source: dw.com
Source: dw.com
The DNA in the adenovirus is modified so that it produces a key part of the SARS-CoV-2 virus particle to which the body then develops an immune response. Johnson Johnson Statement on COVID-19 Vaccine. CNN - Many of the people who got a single dose of the Johnson Johnson COVID-19 vaccine. JNJ is trailing the leaders in the coronavirus vaccine race but the healthcare giant is approaching the finish line thanks to cutting the size of its phase 3 clinical. The agency reviewed clinical trial data from over 40000 participants as well as manufacturing information that Johnson and Johnson provided.
 Source: npr.org
Source: npr.org
Early Johnson Johnson COVID-19 vaccine trial data shows antibody response after 8 days. Our knowledge and experience successes and set-backs alike inform everything were doing today to help eradicate COVID-19. The DNA in the adenovirus is modified so that it produces a key part of the SARS-CoV-2 virus particle to which the body then develops an immune response. The COVID-19 vaccine from Johnson Johnson uses existing technology that involves a virus called adenovirus a common cause of respiratory infections. The agency reviewed clinical trial data from over 40000 participants as well as manufacturing information that Johnson and Johnson provided.
 Source: irishtimes.com
Source: irishtimes.com
Johnson Johnsons Phase III double-dose Covid-19 vaccine prompted a range of expert analyses as to how the prime-boost regimen would be rolled out considering the single-dose approach is already available in the US. The JJJanssen COVID-19 Vaccine was 663 effective in clinical trials efficacy at preventing laboratory-confirmed COVID-19 infection in people who received the vaccine and had no evidence of being previously infected. Johnson Johnson Statement on COVID-19 Vaccine. It took AstraZeneca researchers 1 month to get vaccine trial data to FDA. A clinical trial volunteer participates in Johnson Johnsons study to test a coronavirus vaccine.
 Source: jnj.com
Source: jnj.com
A doctor administers AstraZenecas coronavirus vaccine as part of a clinical trial in São Paulo Brazil on July 24 2020. A clinical trial volunteer participates in Johnson Johnsons study to test a coronavirus vaccine. Johnson Johnson Covid-19 vaccine is 66 effective in global trial but 85 effective against severe disease company says. Johnson Johnson Statement on COVID-19 Vaccine. JNJ is trailing the leaders in the coronavirus vaccine race but the healthcare giant is approaching the finish line thanks to cutting the size of its phase 3 clinical.
This site is an open community for users to share their favorite wallpapers on the internet, all images or pictures in this website are for personal wallpaper use only, it is stricly prohibited to use this wallpaper for commercial purposes, if you are the author and find this image is shared without your permission, please kindly raise a DMCA report to Us.
If you find this site helpful, please support us by sharing this posts to your preference social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title johnson and johnson vaccine trial by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.
Category
Related By Category
- Child tax credit payments bill information
- Child tax credit july 2021 portal information
- Finance of america stock information
- Team usa in olympics information
- Mel gibson christ movie information
- Child tax credit portal down information
- Stephen a smith i dont care gif information
- Usa basketball in olympics information
- Social security yuba city information
- Stephen a smith megan olivi information