Johnson and johnson vaccine kids information
Home » » Johnson and johnson vaccine kids informationYour Johnson and johnson vaccine kids images are available in this site. Johnson and johnson vaccine kids are a topic that is being searched for and liked by netizens today. You can Download the Johnson and johnson vaccine kids files here. Download all royalty-free photos and vectors.
If you’re looking for johnson and johnson vaccine kids images information related to the johnson and johnson vaccine kids topic, you have come to the ideal blog. Our site always gives you suggestions for viewing the highest quality video and image content, please kindly surf and find more enlightening video articles and graphics that fit your interests.
Johnson And Johnson Vaccine Kids. But they will monitor safety efficacy and their timeline may be different than the Pfizer timeline. Johnson Johnson will first test its vaccine in children older than 12 and under 18 but plans to immediately after begin a study that includes newborns and. New Brunswick NJ April 2 2021 Johnson Johnson the Company has begun vaccinating adolescent participants in the ongoing Phase 2a clinical trial for its COVID-19 vaccine candidate developed by the Janssen Pharmaceutical Companies of Johnson Johnson. The Johnson and Johnson COVID-19 vaccine is the third COVID-19 vaccine that the Food and Drug Administration FDA authorized for emergency use.
 Johnson Johnson Tosses 15 Million Doses Pauses Shipments From nymag.com
Johnson Johnson Tosses 15 Million Doses Pauses Shipments From nymag.com
Johnson Johnsons Janssen JJJanssen COVID-19 Vaccine. The drugmaker has been conducting a clinical trial looking at the safety and immune. The Centers for Disease Control and Prevention CDC and the US Food and Drug Administration FDA recommended that use of JJJanssen COVID-19 Vaccine resume in the United States effective April 23 2021. Children 16 and older can receive the Pfizer vaccine. The other two vaccines in use across the US Moderna and Johnson Johnson have both begun clinical trials in children but of the two only Moderna is currently testing children under 18. After initial data from the older teens are reviewed the trial will expand to add adolescents ages 12 to 15.
Children 16 and older can receive the Pfizer vaccine.
Johnson Johnson is now testing its COVID-19 vaccine on teenagers the pharmaceutical company said on Friday. NEW YORK Reuters - Johnson Johnson plans to start testing its experimental COVID-19 vaccine in youths aged 12 to 18 as soon as possible and. Gorsky said during a Washington Post Live event on Wednesday that Johnson Johnson will likely have a COVID vaccine for children by September. The Johnson and Johnson COVID-19 vaccine is the third COVID-19 vaccine that the Food and Drug Administration FDA authorized for emergency use. But they will monitor safety efficacy and their timeline may be different than the Pfizer timeline. The other two vaccines in use across the US Moderna and Johnson Johnson have both begun clinical trials in children but of the two only Moderna is currently testing children under 18.
 Source: statnews.com
Source: statnews.com
The COVID-19 pandemic has had a profound impact on adolescents not just with the. After initial data from the older teens are reviewed the trial will expand to add adolescents ages 12 to 15. Moderna is testing its vaccine. The Centers for Disease Control and Prevention CDC and the US Food and Drug Administration FDA recommended that use of JJJanssen COVID-19 Vaccine resume in the United States effective April 23 2021. But they will monitor safety efficacy and their timeline may be different than the Pfizer timeline.
 Source: nymag.com
Source: nymag.com
The Johnson and Johnson COVID-19 vaccine is the third COVID-19 vaccine that the Food and Drug Administration FDA authorized for emergency use. Moderna is testing its vaccine. Johnson Johnsons single-shot vaccine is currently approved for emergency use in the US. The other two vaccines in use across the US Moderna and Johnson Johnson have both begun clinical trials in children but of the two only Moderna is currently testing children under 18. Children under age 18 were not included in the JJ and Moderna trials and are not eligible for vaccination.
 Source: jnj.com
Source: jnj.com
Pfizer said on Wednesday its two-dose vaccine is 100-percent effective in kids ages 12 to 15. Pfizer and Moderna studies in children are underway. Johnson Johnsons single-shot vaccine is currently approved for emergency use in the US. New Brunswick NJ April 2 2021 Johnson Johnson the Company has begun vaccinating adolescent participants in the ongoing Phase 2a clinical trial for its COVID-19 vaccine candidate developed by the Janssen Pharmaceutical Companies of Johnson Johnson. Johnson Johnson testing COVID vaccine in teenagers between 12 and 17 years old.
 Source: jnj.com
Source: jnj.com
For anyone 18 and older. The Johnson and Johnson COVID-19 vaccine is the third COVID-19 vaccine that the Food and Drug Administration FDA authorized for emergency use. Children 16 and older can receive the Pfizer vaccine. Johnson Johnson testing COVID vaccine in teenagers between 12 and 17 years old. In May children ages 12 to 15 became eligible for the Pfizer shot.
 Source: scdhec.gov
Source: scdhec.gov
New Brunswick NJ April 2 2021 Johnson Johnson the Company has begun vaccinating adolescent participants in the ongoing Phase 2a clinical trial for its COVID-19 vaccine candidate developed by the Janssen Pharmaceutical Companies of Johnson Johnson. The clotting disorder TTS thats believed to be associated with the Johnson Johnson vaccine is a combination of blood clots and thrombocytopenia or low platelets. In May children ages 12 to 15 became eligible for the Pfizer shot. JJ is planning to study the vaccine in children younger than 18. However women younger than 50 years old should especially be aware of the rare risk of blood clots with low platelets after vaccination.
 Source: jnj.com
Source: jnj.com
New Brunswick NJ April 2 2021 Johnson Johnson the Company has begun vaccinating adolescent participants in the ongoing Phase 2a clinical trial for its COVID-19 vaccine candidate developed by the Janssen Pharmaceutical Companies of Johnson Johnson. JJ is planning to study the vaccine in children younger than 18. Children under age 18 were not included in the JJ and Moderna trials and are not eligible for vaccination. Meanwhile Johnson and Johnson has started testing its COVID-19 vaccine on adolescents beginning with those ages 16 and 17. New Brunswick NJ April 2 2021 Johnson Johnson the Company has begun vaccinating adolescent participants in the ongoing Phase 2a clinical trial for its COVID-19 vaccine candidate developed by the Janssen Pharmaceutical Companies of Johnson Johnson.
 Source: standard.co.uk
Source: standard.co.uk
NEW YORK Reuters - Johnson Johnson plans to start testing its experimental COVID-19 vaccine in youths aged 12 to 18 as soon as possible and. In May children ages 12 to 15 became eligible for the Pfizer shot. Pfizer and Moderna studies in children are underway. Pfizer said on Wednesday its two-dose vaccine is 100-percent effective in kids ages 12 to 15. The COVID-19 pandemic has had a profound impact on adolescents not just with the.
 Source: wbur.org
Source: wbur.org
However women younger than 50 years old should especially be aware of the rare risk of blood clots with low platelets after vaccination. In May children ages 12 to 15 became eligible for the Pfizer shot. The other two vaccines in use across the US Moderna and Johnson Johnson have both begun clinical trials in children but of the two only Moderna is currently testing children under 18. Two other vaccines are likely to be approved for children in the coming months. However women younger than 50 years old should especially be aware of the rare risk of blood clots with low platelets after vaccination.
 Source: chla.org
Source: chla.org
Pfizer said on Wednesday its two-dose vaccine is 100-percent effective in kids ages 12 to 15. But they will monitor safety efficacy and their timeline may be different than the Pfizer timeline. Children under age 18 were not included in the JJ and Moderna trials and are not eligible for vaccination. However women younger than 50 years old should especially be aware of the rare risk of blood clots with low platelets after vaccination. Johnson Johnsons Janssen JJJanssen COVID-19 Vaccine.
 Source: abc7news.com
Source: abc7news.com
Meanwhile Johnson and Johnson has started testing its COVID-19 vaccine on adolescents beginning with those ages 16 and 17. Children under age 18 were not included in the JJ and Moderna trials and are not eligible for vaccination. The Johnson and Johnson COVID-19 vaccine is the third COVID-19 vaccine that the Food and Drug Administration FDA authorized for emergency use. Gorsky said during a Washington Post Live event on Wednesday that Johnson Johnson will likely have a COVID vaccine for children by September. The COVID-19 pandemic has had a profound impact on adolescents not just with the.
 Source: washingtonpost.com
Source: washingtonpost.com
The other two vaccines in use across the US Moderna and Johnson Johnson have both begun clinical trials in children but of the two only Moderna is currently testing children under 18. The other two vaccines in use across the US Moderna and Johnson Johnson have both begun clinical trials in children but of the two only Moderna is currently testing children under 18. Pfizer said on Wednesday its two-dose vaccine is 100-percent effective in kids ages 12 to 15. Johnson Johnsons Janssen JJJanssen COVID-19 Vaccine. But they will monitor safety efficacy and their timeline may be different than the Pfizer timeline.
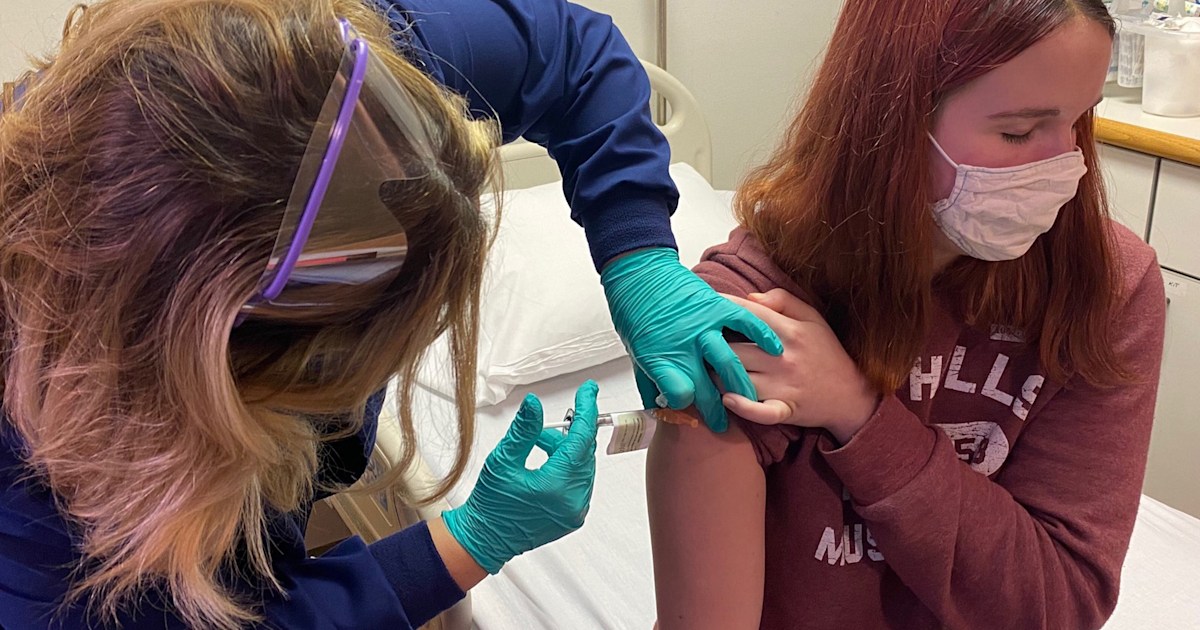 Source: today.com
Source: today.com
After initial data from the older teens are reviewed the trial will expand to add adolescents ages 12 to 15. The Johnson and Johnson COVID-19 vaccine is the third COVID-19 vaccine that the Food and Drug Administration FDA authorized for emergency use. Johnson Johnson has expanded a clinical trial of its experimental coronavirus vaccine to include adolescents 12. However women younger than 50 years old should especially be aware of the rare risk of blood clots with low platelets after vaccination. The other two vaccines in use across the US Moderna and Johnson Johnson have both begun clinical trials in children but of the two only Moderna is currently testing children under 18.
 Source: cincinnatichildrens.org
Source: cincinnatichildrens.org
But they will monitor safety efficacy and their timeline may be different than the Pfizer timeline. Its vaccine is approved for emergency use in anyone age 16 or older. Johnson Johnson plans to initiate its first study of its COVID-19 vaccine involving adolescents ages 12-17 in the fall an executive said Wednesday during a. For anyone 18 and older. But they will monitor safety efficacy and their timeline may be different than the Pfizer timeline.
 Source: thehill.com
Source: thehill.com
Children 16 and older can receive the Pfizer vaccine. Johnson Johnsons Janssen JJJanssen COVID-19 Vaccine. JJ is planning to study the vaccine in children younger than 18. New Brunswick NJ April 2 2021 Johnson Johnson the Company has begun vaccinating adolescent participants in the ongoing Phase 2a clinical trial for its COVID-19 vaccine candidate developed by the Janssen Pharmaceutical Companies of Johnson Johnson. JJ says the first teens are being enrolled in the United Kingdom and Spain.
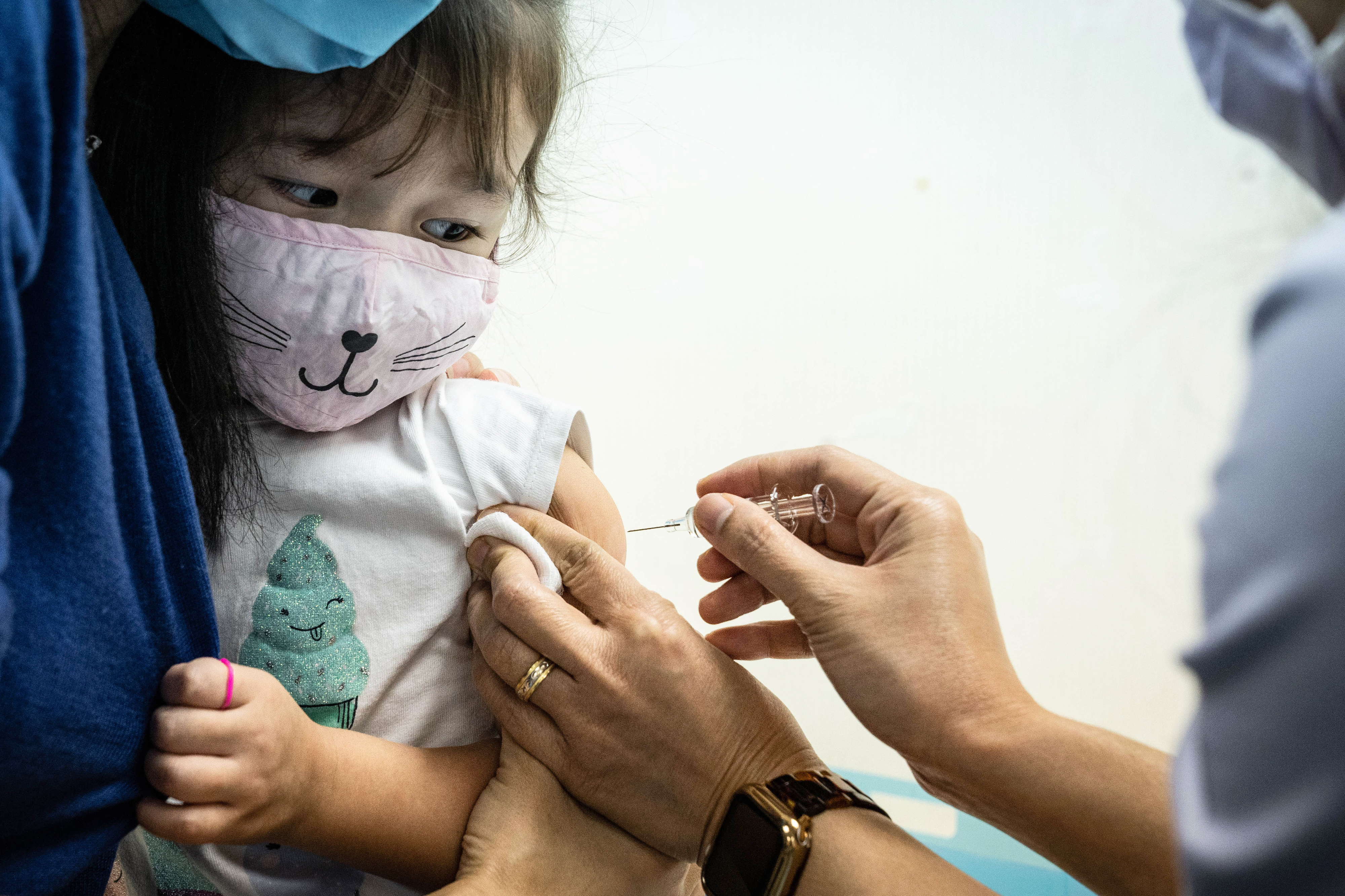 Source: washingtonpost.com
Source: washingtonpost.com
But they will monitor safety efficacy and their timeline may be different than the Pfizer timeline. Johnson Johnson is now testing its COVID-19 vaccine on teenagers the pharmaceutical company said on Friday. New Brunswick NJ April 2 2021 Johnson Johnson the Company has begun vaccinating adolescent participants in the ongoing Phase 2a clinical trial for its COVID-19 vaccine candidate developed by the Janssen Pharmaceutical Companies of Johnson Johnson. The other two vaccines in use across the US Moderna and Johnson Johnson have both begun clinical trials in children but of the two only Moderna is currently testing children under 18. JJ says the first teens are being enrolled in the United Kingdom and Spain.
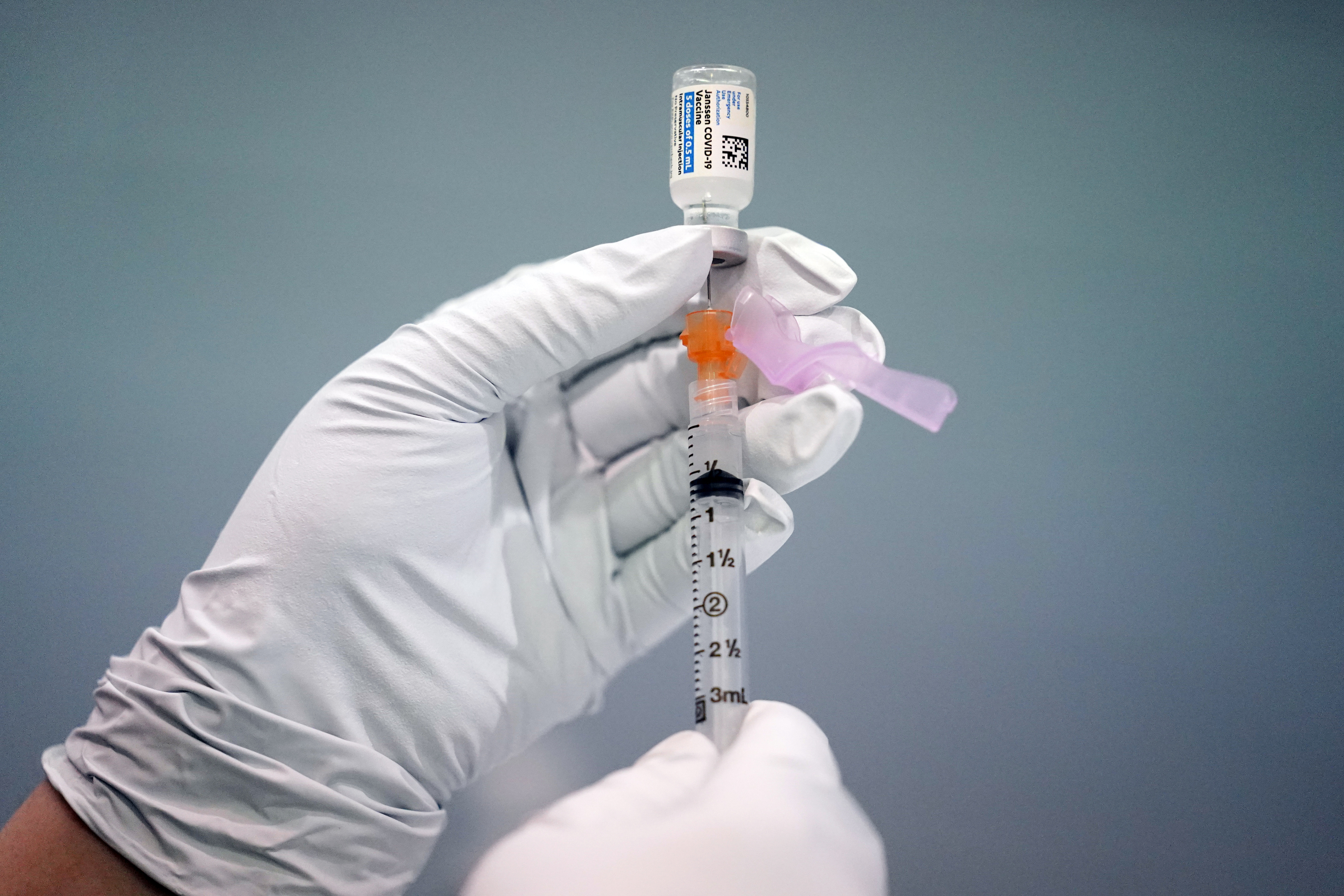 Source: washingtonpost.com
Source: washingtonpost.com
After initial data from the older teens are reviewed the trial will expand to add adolescents ages 12 to 15. Johnson Johnson plans to initiate its first study of its COVID-19 vaccine involving adolescents ages 12-17 in the fall an executive said Wednesday during a. In May children ages 12 to 15 became eligible for the Pfizer shot. Its vaccine is approved for emergency use in anyone age 16 or older. Pfizer said on Wednesday its two-dose vaccine is 100-percent effective in kids ages 12 to 15.
 Source: fox8.com
Source: fox8.com
Johnson Johnsons Janssen JJJanssen COVID-19 Vaccine. Pfizer said on Wednesday its two-dose vaccine is 100-percent effective in kids ages 12 to 15. Meanwhile Johnson and Johnson has started testing its COVID-19 vaccine on adolescents beginning with those ages 16 and 17. The Johnson and Johnson COVID-19 vaccine is the third COVID-19 vaccine that the Food and Drug Administration FDA authorized for emergency use. Two other vaccines are likely to be approved for children in the coming months.
 Source: pittwire.pitt.edu
Source: pittwire.pitt.edu
For anyone 18 and older. Pfizer and Moderna studies in children are underway. The other two vaccines in use across the US Moderna and Johnson Johnson have both begun clinical trials in children but of the two only Moderna is currently testing children under 18. But they will monitor safety efficacy and their timeline may be different than the Pfizer timeline. JJ says the first teens are being enrolled in the United Kingdom and Spain.
This site is an open community for users to do sharing their favorite wallpapers on the internet, all images or pictures in this website are for personal wallpaper use only, it is stricly prohibited to use this wallpaper for commercial purposes, if you are the author and find this image is shared without your permission, please kindly raise a DMCA report to Us.
If you find this site value, please support us by sharing this posts to your preference social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title johnson and johnson vaccine kids by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.
Category
Related By Category
- Child tax credit payments bill information
- Child tax credit july 2021 portal information
- Finance of america stock information
- Team usa in olympics information
- Mel gibson christ movie information
- Child tax credit portal down information
- Stephen a smith i dont care gif information
- Usa basketball in olympics information
- Social security yuba city information
- Stephen a smith megan olivi information